BACKGROUND
Severe scabies, a rare parasitic skin disease characterized by abundant skin mites, may be life-threatening and poses public health concerns worldwide. A combination of standard-dose oral ivermectin and topical scabicides is recommended for treatment. However, data from randomized clinical trials are lacking, and the probability of cure is uncertain. Ivermectin at higher doses has been effective in the treatment of some parasitic diseases.
METHODS
We conducted a blinded randomized trial involving adults with severe scabies (i.e., profuse or crusted), as confirmed by parasitologic or dermoscopic assessment. The patients were assigned in a 1:1 ratio to receive oral ivermectin (to be taken with food) at a dose of 400 μg per kilogram of body weight (higher-dose group) or 200 μg per kilogram (standard-dose group) on days 0, 7, and 14, combined with head-to-toe application of 5% permethrin cream on days 0 and 7 and daily application (as recommended) of an emollient cream. The primary end point was cure of severe scabies, which was defined as the absence of mites and mite-related products (i.e., eggs and feces), as confirmed by parasitologic or dermoscopic assessment on days 18 and 21, and the absence of active clinical lesions on physical examination on day 28.
RESULTS
A total of 132 patients (66 in each group) were included in the main analysis. Cure was observed in 75% of the patients in the higher-dose group and in 82% of those in the standard-dose group (odds ratio for cure, 0.64; 95% confidence interval, 0.25 to 1.67). No safety issues were identified.
CONCLUSIONS
Among adults, the 400-μg-per-kilogram dose of ivermectin plus 5% permethrin cream was not superior to the standard 200-μg-per-kilogram dose of ivermectin plus 5% permethrin cream in curing severe scabies. (Funded by the French Ministry of Health and French Society of Dermatology; ClinicalTrials.gov number, NCT02841215.)
Severe scabies is a rare parasitic skin disease that encompasses profuse and diffuse manifestations of classic scabies and crusted scabies.1,2 Clinically, the rash in profuse scabies is erythematous, scaly, and disseminated, involving areas that are not typically affected in classic scabies (e.g., the scalp, head, neck, or back) and potentially engendering erythroderma. Crusted scabies is clinically characterized by the presence of several hyperkeratotic and psoriasiform skin lesions containing dozens to hundreds to thousands of infesting Sarcoptes scabiei var. hominis mites. Immunosuppression — either iatrogenic (e.g., caused by systemic or topical use of glucocorticoids in patients with transplants or in those with unrecognized scabies-related itch or eczematous lesions, respectively) or noniatrogenic (e.g., caused by underlying human immunodeficiency virus, acquired immunodeficiency syndrome, or human T-cell leukemia virus type 1) — is a risk factor for severe scabies. At-risk subpopulations include, for example, older adults, Indigenous communities, and migrants, especially those living in institutions or overcrowded settings.3 Severe scabies may be life-threatening, especially for patients with coexisting conditions. The condition causes fluid and electrolyte disturbances, bacteremia, necrotizing soft-tissue infections, and fatal sepsis.4,5 At the public health level, severe scabies is a major concern worldwide. The disease is highly contagious and can lead to subsequent outbreaks, such as in hospitals, nursing homes, prisons, shelters, and communities with poor health resources, and to high levels of endemicity in low- and middle-income countries, because patients with crusted scabies are sources of reinfestation after intervention programs.6,7
Regardless of the disease context, such as immunosuppression, severe scabies is refractory to the usual treatment for classic scabies (one scabicide taken twice, 7 to 14 days apart) owing to the abundance of skin mites. Multiple methods of treatment are necessary and may include hospitalization, room isolation, the use of protective garments, environmental disinfection, and, most important, a repeated combination of oral scabicides (e.g., ivermectin) and topical scabicides (e.g., 5% permethrin).8,9 In fact, on the basis of clinical experience, case series, and expert recommendations and guidelines, several regimens are prescribed, which results in cure in 30 to 100% of cases.10,11 This broad range of uncertainty reflects the retrospective design of studies, small sample sizes, and the lack of randomized clinical trials.
Because ivermectin at higher doses has been effective in the treatment of some parasitic diseases, such as head lice and malaria,12,13 we conducted a randomized clinical trial involving adults with severe scabies to compare the efficacy and safety of a three-dose regimen of oral ivermectin at a higher dose (400 μg per kilogram of body weight) with those of the standard dose (200 μg per kilogram), combined with two applications of 5% permethrin cream.
Methods
TRIAL DESIGN AND OVERSIGHT
The GALE CRUSTED (High Dosage of Oral Ivermectin for Crusted Scabies) trial was a blinded, head-to-head randomized clinical trial that evaluated whether oral ivermectin at a high dose was superior to the standard dose in curing severe scabies; with the patients in both groups receiving 5% permethrin and emollient cream. The protocol and statistical analysis plan are available with the full text of this article at NEJM.org. A statistician, who was not involved in patient recruitment, prepared the 1:1 randomization list in blocks of four. Randomization was performed without stratification with the use of a secure, centralized, interactive, Web-based response system.
Patients were assigned to receive oral ivermectin (to be taken with food) at a dose of 400 μg per kilogram or at the standard 200-μg-per-kilogram dose on days 0, 7, and 14. Because the pharmacist prepared the same number of matched tablets and delivered identical unmarked wallets to both trial groups, the patients and investigators remained unaware of the trial-group assignments. Table S2 in the Supplementary Appendix, available at NEJM.org, shows the number of tablets administered to the patients according to body weight and describes the organization of unmarked wallets and evaluation of adherence. On days 0 and 7, the patients in both treatment groups also applied 5% permethrin cream from head to toe in a quantity sufficient to cover the entire skin surface, including the hair, face, and genitalia; the cream was left on overnight (≥8 hours) and washed off in the morning. Daily application of emollient cream was recommended for all the patients (a topical keratolytic agent [i.e., 5% salicylic acid in petroleum jelly] was permitted for crusted scabies). The trial timeline is shown in Figure S1. Patients could be hospitalized and isolated if necessary, and permethrin could be applied by a dedicated nurse if needed. The measures to prevent scabies from spreading within patients’ households were organized by the investigators according to local guidelines.
The trial protocol was approved by a French National Ethics Committee (Comités de Protection des Personnes Île de France). All the patients or their legally authorized representatives provided written informed consent. The authors vouch for the accuracy and completeness of the data and for the fidelity of the trial to the protocol.
PATIENTS AND ELIGIBILITY CRITERIA
Adults 18 years of age or older with or without immunosuppression (including those with immunosuppression who had previously received topical or systemic glucocorticoids) were eligible to participate if they had severe scabies — either profuse (defined as spreading, erythematous, scaly lesions) or crusted (with at least two hyperkeratotic sites).1,2 All clinical cases were confirmed by diagnostic parasitologic examination of skin scrapings with direct microscopy to visualize the presence of mites, mites in immature stages, or mite-related products (i.e., eggs and feces) or by dermoscopic assessment to visualize the specific “delta-wing jet” pattern.14,15 Patients who had taken antiparasitic ivermectin or albendazole within the past 7 days were excluded, but those who had been previously treated with topical scabicides were not.
END POINTS
The primary end point was cure of severe scabies, which was defined as the absence of mites and mite-related products, as confirmed by parasitologic or dermoscopic assessment (or both) on days 18 and 21 (a single positive result was considered to indicate treatment failure) and the absence of active clinical scabies lesions on physical examination on day 28. In post hoc sensitivity analyses, we considered a patient to be cured if there was at least one parasitologic or dermoscopic assessment that showed no visual evidence of mites or mite-related products on day 18 or day 21 (taking into account that not all the patients would have assessments on both day 18 and day 21), no active lesions on physical examination on day 28, and no receipt of rescue therapy, which would result in an outcome being reclassified as treatment failure. The secondary end point was the incidence of adverse events. Data on adverse events were collected by the investigators during each patient visit or telephone call.
STATISTICAL ANALYSES
We calculated that a sample of 130 patients would provide the trial with 90% power to detect a between-group difference of 20 percentage points in the percentage of patients with cure of scabies (i.e., 95% in the higher-dose group and 75% in the standard-dose group) at a two-sided type I error of 5%. Patients were evaluated according to their treatment assignment; those who did not receive any drug were excluded from the statistical analysis. Because the trial was blinded, “no drug intake” was considered to be independent of the treatment group, and the exclusion of these patients did not introduce bias in the estimation of the treatment effect.16
The characteristics of the patients at the time of inclusion are reported descriptively for each trial group. Missing values were managed with the use of multiple imputation. We analyzed the primary end point by fitting a logistic model to estimate the odds ratio for cure, with an odds ratio greater than 1 indicating more favorable results in the higher-dose group than in the standard-dose group. An analysis that included only patients with complete data for the primary end point (i.e., those who had undergone assessments of mite and mite-related products on days 18 and 21 and physical examination on day 28) was also performed. As complementary analyses, two other types of models were fitted: linear models with the identity-link functions, which allow estimating risk differences, and log-linear models with log-link functions, which allow estimating relative risks (see the Supplementary Appendix). Post hoc subgroup analyses were also performed according to the use of immunosuppressive drugs (yes or no), scabies type (crusted or profuse only), and time since
PATIENT CHARACTERISTICS
The trial started on October 11, 2017, and ended on January 25, 2022. A total of 153 patients were screened at 33 dermatology departments in France, and 133 underwent randomization — 66 were assigned to the higher-dose ivermectin group, and 67 to the standard-dose ivermectin group. One patient in the standard-dose group received no treatment and was excluded from further statistical analyses. A total of 47 patients in the higher-dose group and 50 of those in the standard-dose group completed the trial, with parasitologic or dermoscopic assessment (or both) on days 18 and 21 and physical examination on day 28. In the sensitivity analyses, outcomes were known for 53 patients who received the higher dose of ivermectin and for 55 of those who received the standard dose (Fig. S2). In total, 52 of 66 (79%) patients in the higher-dose group and 54 of 66 patients (82%) in the standard-dose group returned the wallets, which showed that adherence was good, with 45 patients (87%) patients and 43 patients (80%), respectively, completing the assigned regimen.
The median age of the patients across treatment groups was 67 years (Table 1), with one third of the patients being older than 80 years. Nearly 60% of the patients were hospitalized, and 42% had neurologic or cognitive impairment. Among all the patients, 59% had profuse scabies only, 5% had crusted scabies only, and 36% had both types. The median time since the onset of signs or symptoms of scabies was 81 days (interquartile range, 42 to 126) in the higher-dose group and 109 days (interquartile range, 58 to 224) in the standard-dose group. Diagnostic dermoscopy alone confirmed mite infestation in approximately 70% of the patients (Table S3). The patients were representative of the general population with severe scabies (Table S4).
TABLE 1
Demographic and Clinical Characteristics of the Patients at the Time of Inclusion.
CURE
The results of the main analysis showed no significant difference in the percentage of patients with cure between the higher-dose group and the standard-dose group on day 28 (75% vs. 82%; odds ratio of cure, 0.64; 95% confidence interval [CI], 0.25 to 1.67) (Table 2). In the sensitivity-analysis population, 11 patients received rescue therapy (6 in the higher-dose group and 5 in the standard-dose group), which led to the outcome being reclassified as treatment failure in 4 patients (1 in the higher-dose group and 3 in the standard-dose group) — 7 of these patients were already considered to have treatment failure. The sensitivity analysis also showed no between-group difference in the percentage of patients with cure (82% vs. 83%; odds ratio of cure, 0.91; 95% CI, 0.35 to 2.35). The results of the analyses in which only patients with complete data were considered were in agreement with those in the main analysis. The results of post hoc subgroup analyses are shown in Table 3. In the subgroup analysis according to scabies type, the odds ratio for cure in the higher-dose group, as compared with the standard-dose group, was 0.34 (95% CI, 0.08 to 1.39) among the patients with crusted scabies and 2.14 (95% CI, 0.38 to 12.06) among those with profuse scabies only.
TABLE 2
Patients with Cure on Day 28.
TABLE 3
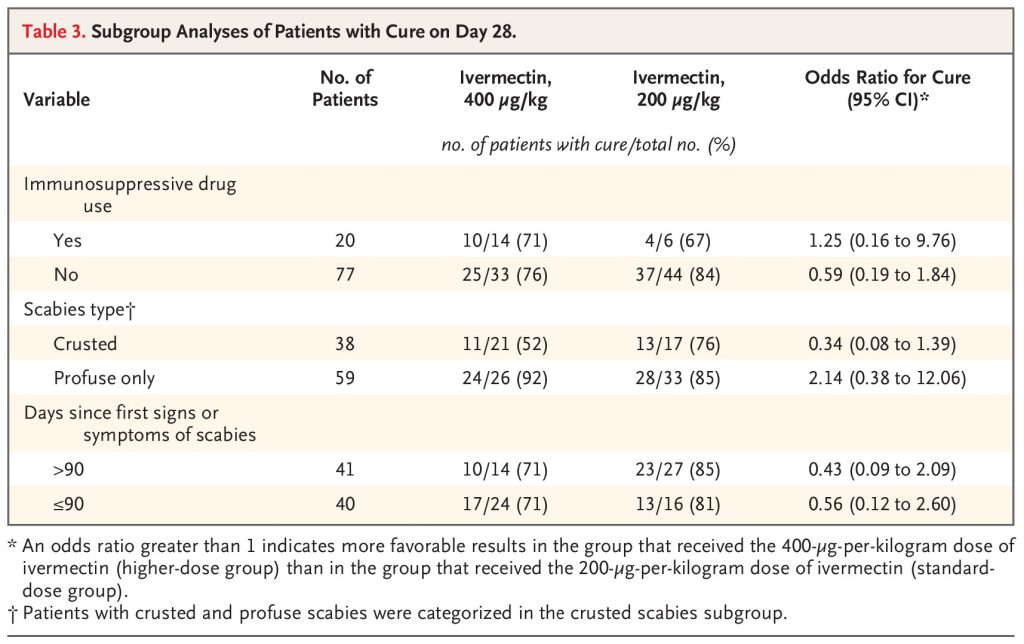
Subgroup Analyses of Patients with Cure on Day 28.
SAFETY
Serious adverse events occurred in 8 patients — 3 in the higher-dose group and 5 in the standard-dose group. In the higher-dose group, one serious adverse event occurred (septicemia due to cutaneous infection) and was deemed by the trial investigator to be probably related to the trial drug, and 2 patients died from cardiac decompensation. In the standard-dose group, two serious adverse events occurred that were deemed by the trial investigator to be probably related to the trial drug (prolonged hospitalization for eczema and for septicemia in 1 patient each), and 2 patients died from intercurrent diseases (1 from neurologic failure and 1 from cardiac failure) and 1 from hemorrhagic failure. At least one cutaneous adverse event that was attributed by the investigator to be potentially related to the prescribed scabicide combination occurred in 14 patients in each treatment group; these events mainly included eczematous or contact dermatitis, erythema or edema, and itching or burning (Table 4).
TABLE 4
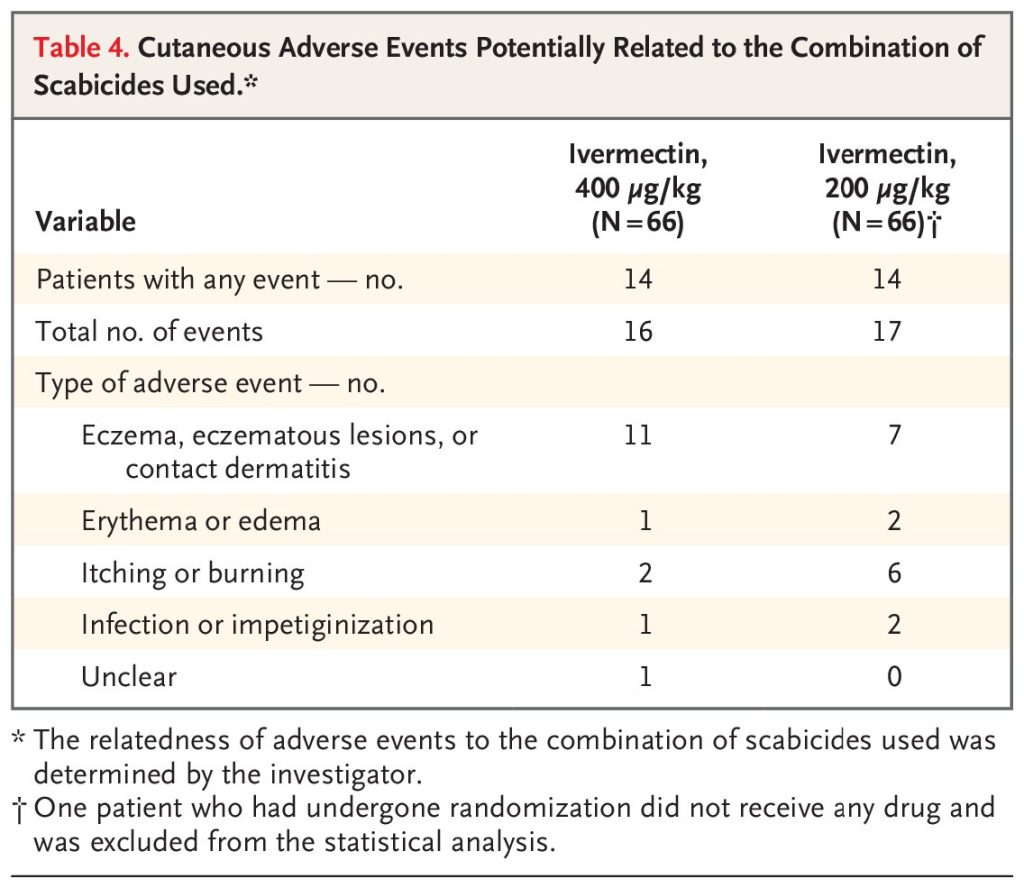
Cutaneous Adverse Events Potentially Related to the Combination of Scabicides Used.
Discussion
Although high numbers of parasites are characteristic of severe scabies, we found no evidence that the probability of cure was higher with the 400-μg-per-kilogram dose of ivermectin (taken with food on days 0, 7, and 14) than with the standard 200-μg-per-kilogram dose, when treatment was combined with head-to-toe application of 5% permethrin cream on days 0 and 7 and daily use of emollient cream. On day 28, cure was observed in 75% of the patients in the higher-dose group and in 82% of those in the standard-dose group. Adherence to ivermectin was good, and we assumed that the protocol-directed care administered in our trial was correct in such a patient population with a severe form of scabies, among whom specialized care and hospitalization is often indicated.
Unlike other parasites (e.g., head lice) that are blood-sucking arthropods, S. scabiei mites ingest intercellular fluid (lymph) and are thus potentially less targetable by oral ivermectin, even if an adequate plasma drug concentration is attained.12,17 Beyond showing that a higher dose of ivermectin was no better than the standard dose in curing severe scabies, the results of our trial show that repeated administration of oral ivermectin at a dose of 200 μg per kilogram, combined with topical application of 5% permethrin and an emollient cream (with or without keratolytic agents), could be used for profuse and crusted scabies.
We anticipate good real-world applicability of our results. In our trial, all clinical cases of scabies, either profuse or crusted, were confirmed at the time of inclusion by adequate diagnostic parasitologic or dermoscopic assessment, which are the currently validated methods for scabies studies.18 The 133 patients enrolled in the trial adequately represented the adult population with severe scabies11: the patients were generally older than those with classic scabies, the profuse type of severe scabies was more prevalent than the crusted type, more than 40% of the patients had neurologic or cognitive impairment, and the possible place of contamination was more likely to be an institutional health care setting (e.g., hospital or nursing home). The prevalence of iatrogenic immunosuppression was probably underestimated because only the drugs that were being taken at the time of inclusion — and not those that were previously prescribed — were recorded. Moreover, the patients had had signs or symptoms of scabies for several weeks, and at-risk treatments might have been stopped before inclusion.
The use of wallets that were prepared in advance ensured that the patients and investigators were unaware of the trial-group assignments, thereby avoiding bias in outcome measurements. Because of the lack of standardized international consensus guidelines for the management of crusted scabies, as well as the lack of trial data on outcomes,2 we used our 2014 protocol definition of cure as a primary end point — namely, the absence of mites and mite-related products, as confirmed on parasitologic or dermoscopic assessment on days 18 and 21, and clinical cure of scabies (i.e., the absence of active clinical scabies lesions on physical examination) on day 28. With regard to the sensitivity analysis, visual confirmation of the absence of mites and mite-related products by parasitologic or dermoscopic assessment on day 18 or day 21, clinical cure on day 28, and no receipt of rescue therapy, the percentage of patients with cure did not differ meaningfully between the trial groups, a finding that reinforces the consistency of the results. Indeed, any recourse to rescue therapy was considered to be a treatment failure, even when no mites were seen. We acknowledge that mite searches were not done on day 18 and day 21 for 15 patients for several possible reasons: older adults may be reluctant to attend multiple hospital visits, which reflects the known challenge of retaining older patients in clinical trials,19 and the poor feasibility of second mite searches in patients who had a negative result 3 days earlier.
Although important, no strong definition of cured severe scabies exists. Hence, being mite-free at least once on rescraping for parasitologic or dermoscopic reexamination on day 18 or day 21 and the absence of clinical scabies on day 28 may be sufficient in clinical practice for “common” severe scabies.20 Moreover, the results of our subgroup analyses of the treatment effect according to immunosuppressive drug use, scabies type, and time since the onset of signs or symptoms of scabies were similar to those of the main analysis, thereby strengthening our conclusion regarding the nonsuperiority of the higher-dose ivermectin regimen over the standard-dose regimen.
Despite the lack of clear evidence regarding the management of severe scabies, most guidelines have recommended prolonged and repeated therapeutic regimens to ensure complete eradication of the profusion of mites, including those newly hatched and eggs.21,22In the Northern Territory of Australia, which includes socioeconomically disadvantaged Indigenous populations with a high prevalence of infestation, a grading of crusted scabies has been established, and different drug regimens have been prescribed accordingly (e.g., standard-dose oral ivermectin to be taken on 3 days [days 0, 1 and 7], 5 days [plus days 8 and 14], or 7 days [plus days 21 and 28], combined with topical permethrin or benzyl benzoate to be taken every 2 to 3 days for 1 to 2 weeks).23,24 The Centers for Disease Control and Prevention also recommends those protocols, depending on the severity of infection.22Indeed, 5% permethrin cream, a topical scabicide of choice, even showed superiority over oral ivermectin in a cluster randomized clinical trial involving patients with classic scabies.25 In geographic areas where S. scabiei mites are tolerant or resistant to permethrin, other topical scabicides (e.g., 10 to 25% benzyl benzoate emulsion or 1% lindane cream) could be applied in combination therapy, although 1% lindane cream is potentially neurotoxic and was withdrawn from various markets.26-30 Larger randomized trials should be designed to determine whether the 82% probability of cure of severe scabies with standard-dose ivermectin can be improved. Strategies for how to attain higher percentages and potentially faster rates of cure include prolonged administration of oral ivermectin, more frequent applications of possibly different topical scabicides plus keratolytic agents to enhance their penetration into thickened skin, or administration of moxidectin, a macrocyclic lactone with a longer half-life than ivermectin.31
Safety-related outcomes with oral ivermectin were good, with results that were similar in the higher-dose and standard-dose groups, as previously reported.32 Oral ivermectin has been shown to be safe in older adults,33 and no safety concerns were identified among the patients with severe scabies in our trial, one third of whom were older than 80 years of age. The skin of patients with severe scabies is often in poor condition as a result of chronic pruritus and various scabies lesions, dermatoporosis in the oldest patients, and irritant or allergic contact dermatitis related to the use of topical scabicides.34 The relatively low incidence of cutaneous adverse events in our trial is noteworthy and perhaps reflects the daily application of an emollient cream. In addition, 60% of the patients were hospitalized in dermatology departments that had nurses who were dedicated to skin surveillance and care, and as outpatients they received only two permethrin applications.
The strengths of our trial include its randomized design, the blinded assessment of end points, the fact that cure was determined on the basis of clinical cure on day 28 (after one or two previous negative parasitologic or dermoscopic assessments for mites), and the expected good generalizability of the findings, all of which provide confidence in the accuracy of the reported treatment effects. The limitations of this trial include the following: there were no patients with very severe crusted scabies in the trial population; data were unavailable between the time of symptom onset and trial enrollment (including data on previous medications); diagnostic procedures were not standardized, which would have been preferable; adherence to permethrin was not evaluated; some patients did not undergo the two consecutive parasitologic or dermoscopic assessments on days 18 and 21; the trial was located in France, where molecular and clinical tolerance and levels of resistance to permethrin are thought to be low35; there was no long-term follow-up of patients or later contact with them; and transmission was not evaluated.
Although this randomized trial involving patients with severe scabies did not provide evidence that higher-dose ivermectin was superior to the standard dose, the results showed oral ivermectin taken at a dose of 200 μg per kilogram on days 0, 7, and 14, combined with 5% permethrin cream applied on days 0 and 7 and daily application of an emollient cream (with or without keratolytic agents), led to an 82% probability of cure of profuse and crusted scabies on day 28.
NOTES
A data sharing statement provided by the authors is available with the full text of this article at NEJM.org.
Supported by a grant (Programme Hospitalier de Recherche Clinique, PHRCN-14-0612) from the French Ministry of Health and by a grant (AO2019-SRK19008) from the French Society of Dermatology.
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
We thank all the patients, their health care providers, and clinical research assistants; Françoise Botterel, M.D., Ph.D. (Department of Parasitology), Audrey Colin (Department of Dermatology), Hayat Medjenah, M.Sc. (Unité de Recherche Clinique), Bernard Do, Pharm.D., Muriel Paul, Pharm.D., and Olivier Thirion, Pharm.D. (Unité Pharmaceutique de Recherche en Essais Cliniques), all at Hôpital Henri-Mondor, Créteil, France, for their assistance in facilitating the conduct of this trial; Priscilla Andriamiandrisos, Pharm.D., Sarah Dalibey, M.D., Sofiane Mokrani, Pharm.D., Anne Radenne, M.Sc., and Malika Yahmi, M.Sc. (Secteur Vigilance et Pôle Promotion, Direction de la Recherche Clinique et de l’Innovation de l’AP-HP, Paris) for their assistance in the administrative organization of the randomized clinical trial; Aurore Foureau, M.Sc., Center of Evidence of the French Society of Dermatology, for her help in organizing previous versions of the manuscript tables; and Janet Jacobson, M.Sc., for editorial assistance with an earlier version of the manuscript.